 Ī small-scale, nonbiological route from D-mannitol is illustrated in the following scheme: Applications Polymers įorty-five percent of propylene glycol produced is used as a chemical feedstock for the production of unsaturated polyester resins. Three-carbon deoxysugars are also precursor to the 1,2-diol. This conversion is the basis of a potential biotechnological route to the commodity chemical 1,2-propanediol. Dihydroxyacetone phosphate, one of the two products of breakdown ( glycolysis) of fructose 1,6-bisphosphate, is a precursor to methylglyoxal. Lactic acid and lactaldehyde are common intermediates. (S)-Propanediol is synthesized via fermentation methods. This starting material is usually reserved for industrial use because of the noticeable odor and taste that accompanies the final product. Propylene glycol can also be obtained from glycerol, a byproduct from the production of biodiesel. Use of USP (US Pharmacopoeia) propylene glycol can reduce the risk of Abbreviated New Drug Application (ANDA) rejection. Further purification produces finished industrial grade or USP/JP/EP/BP grade propylene glycol that is typically 99.5% or greater. įinal products contain 20% propylene glycol, 1.5% of dipropylene glycol, and small amounts of other polypropylene glycols. Manufacturers use either non-catalytic high-temperature process at 200 ☌ (392 ☏) to 220 ☌ (428 ☏), or a catalytic method, which proceeds at 150 ☌ (302 ☏) to 180 ☌ (356 ☏) in the presence of ion exchange resin or a small amount of sulfuric acid or alkali. According to a 2018 source, 2.16 M tonnes are produced annually. Industrially, propylene glycol is mainly produced from propylene oxide (for food-grade use). The S-isomer is produced by biotechnological routes. Commercial processes typically use the racemate. The compound is sometimes called (alpha) α-propylene glycol to distinguish it from the isomer propane-1,3-diol, known as (beta) β-propylene glycol. Propylene glycol is approved and used as a vehicle for topical, oral, and some intravenous pharmaceutical preparations in the U.S. Propylene glycol is a compound which is GRAS (generally recognized as safe) by the US Food and Drug Administration under 21 CFR x184.1666, and is also approved by the FDA for certain uses as an indirect food additive. Propylene glycol is also present in propylene glycol alginate, which is known as E405. For cosmetics and pharmacology, the number is E490. In the European Union, it has E-number E1520 for food applications. It is produced on a large scale primarily for the production of polymers. 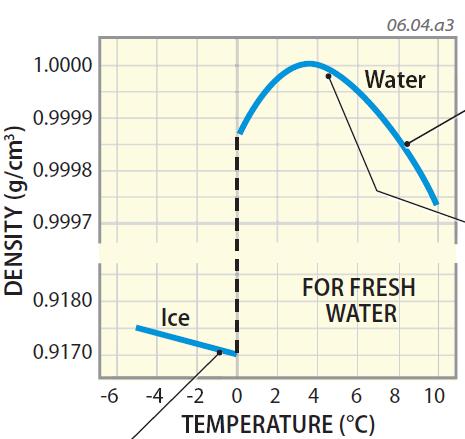
In general, glycols are non-irritating and have very low volatility. It is miscible with a broad range of solvents, including water, acetone, and chloroform. Its chemical formula is CH 3CH(OH)CH 2OH.Īs it contains two alcohol groups, it is classed as a diol. 
Direct measurement for density of seawater is done by maintaining the collected water samples in controlled environmental conditions.Propylene glycol ( IUPAC name: propane-1,2-diol) is a viscous, colorless liquid, which is nearly odorless but possesses a faintly sweet taste. Likewise, check the water density of samples having same salinity and different temperatures. All you need is enter the temperature (both in Celsius and Fahrenheit) and salinity in PPM (parts per million) or mg/L in the suggested fields.Īfter finding the results, you can correlate the density of water at room temperature for different salinity levels. If you are interested in calculating the density of seawater at different temperatures, there are online tools that are developed for the same purpose. Thus, water with lower salt content will float over water with higher salt concentration. In short, density of water is directly proportional to salinity. In general, the density of a matter is higher in solid form, as compared to the density of the same matter in liquid form.Īs with salinity, higher the level of salt concentration, the more will be the density of water, even at room temperature. /experiment-showing-relative-density-of-different-liquids-liquids-of-different-densities-will-sit-on-top-of-each-other-without-mixing-and-if-mixed-together-will-re-settle-into-layers-the-most-dense-liquid-lies-at-the-bottom-the-least-dense-at-the-t-57a768ac3df78cf459161869.jpg)
However, as we are discussing the density of water measured in ground level for collected water samples, the pressure attribute is not considered in determining the density. Of course, pressure does play a role in the subject of water denseness. While discussing this topic in detail, water density is found to vary with respect to the level of salinity (or other impurities) and fluctuation of temperature. However, it is to be noted that grams per milliliter (g/mL) and gram per cubic centimeter (g/cm 3) are not SI units. The logic is to divide the value of kg/m 3 by 1000 to get pure water density in g/ml. In other words, at the same temperature, the density of water in g/ml (or g/cm 3) is 0.99777. 
At room temperature (i.e., 22° C), the density of water in kg/m 3 is 997.77. With respect to the definition, the density formula is represented as Density (ρ) = Mass (m)/Volume (V). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
